 
The etymology of the prefixes derived from Rule A-1.1 is only loosely based on the corresponding Greek words. This appears to be quite a reasonable mnemonic method and if we want to extend the list, we shall need a specific ending for hundreds and for thousands. Nevertheless, in spite of the difficulties inherent to this "inverted" pattern, it is now so much entrenched in common day practice that any alternative order is out of the question.Įxamination of the existing terms shows that names for multiples of tens beyond twenty are formed by adding the ending "-conta-" to the name of the corresponding units, with insertion of an "a" for thirty: The general pattern in which units are cited first, then tens, hundreds, etc., has some disadvantages compared with the natural order of ciphers in arabic numbers, which is used in English and French, but not always in German. However, the multiplying prefixes "bis-", "tris-", etc., are used instead of "di-", "tri-", etc., when the use of the latter would result in a potential ambiguity for example, tris(decyl) is used to describe three decyl substituents because tridecyl is the name for a thirteen carbon acyclic substituent.Īs far as possible, the principles embodied in the present terms have been used to extend the list. Multiplying prefixes are used in much the same way to denote other identical structural features of a chemical compound, such as principal characteristic groups, sites of unsaturation, and number of ionic centers. For substituted substituents, the multiplying prefixes bis-, tris-, and tetrakis- are used higher multiplying prefixes are formed by replacing the "-ne" of the corresponding alkane with "-kis-". For simple substituents, i.e., when not themselves substituted, the multiplying prefixes di-, tri-, and tetra-, are generally used higher multiplying prefixes are formed by suppressing the ending "-ne" of the name of the corresponding alkane. The number of identical substituents to a parent compound is expressed according to Rule A-2.5 ( ref. Until recently, numerical terms higher than 199 have not been required but now, in order to avoid confusion from alternative suggestions, it seems advisable to give a clear method for extending the list. 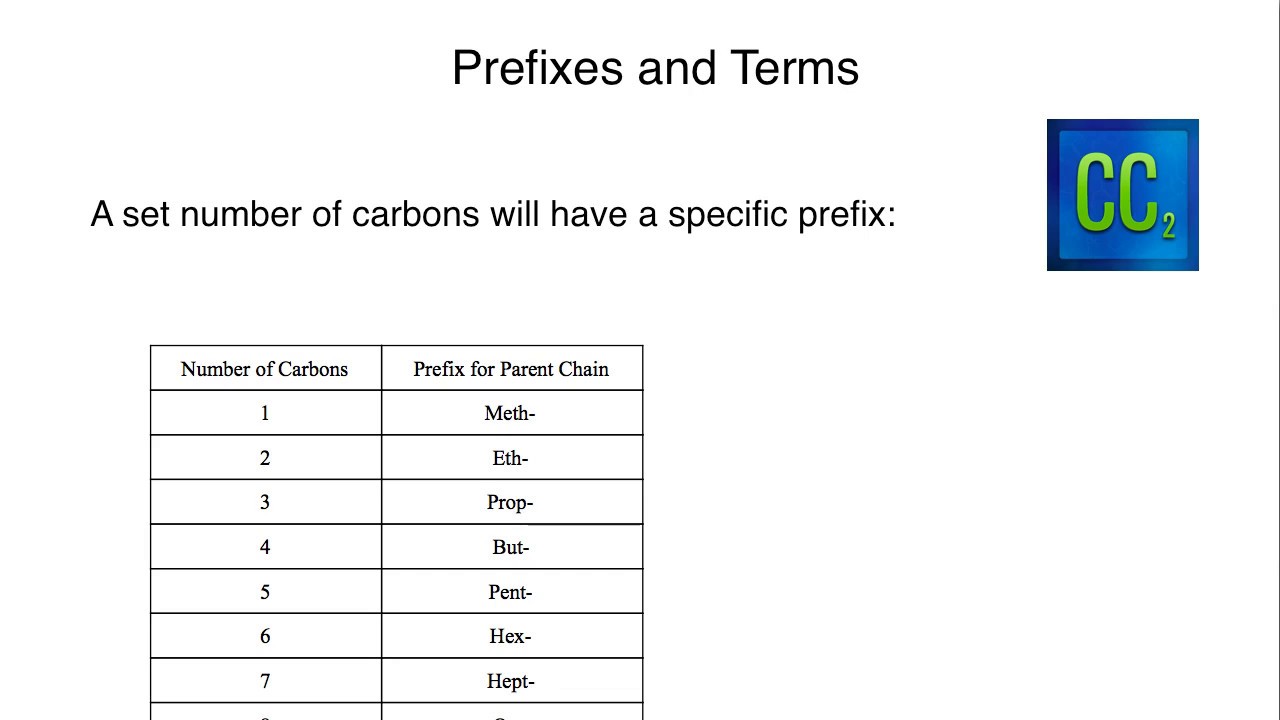
1a) uses numerical terms to name saturated unbranched acyclic hydrocarbons containing up to 199 carbon atoms. This document provides the necessary guidance, based on principles already established, for generating numerical terms up to 9999 by using the infixes "cta" (for the hundreds digits) and "lia" (for the thousands digits) in a way quite analogous to the use of "conta" for the tens digits beyond twenty. Recently a desire has been expressed for numerical terms higher than 199. The use of these numerical terms for expressing identical substituents to a parent structure, and their modifications for use with "complex" substituents, is described in Rule A-2.5. Rule A-1.1 of the IUPAC Organic Nomenclature Rules illustrates the numerical terms through 199 in the names of saturated unbranched acyclic hydrocarbons. Numerical terms are used in chemical names for indicating a number of identical structural units in a compound. NT-2 Numerical terms for complex features Chem., 1989, 43 103-105 (in Polish).įor problems in converting the text into a World Wide Web version see the IUPAC home page.Īny comments, corrections or suggestions for a future edition should be e-mailed to Numerical terms for simple features A PDF of the printed version is available. If you need to cite these rules please quote this reference as their source. Mile End Road, London, E1 4NS, Rules are as close as possible to the published version. School of Physical and Chemical Sciences, Queen Mary University of London, Such acids include sulfuric acid (H 2 SO 4 ) or carbonic acid (H 2 CO 3 ).Numerical Terms International Union of Pure and Applied Chemistry.Ĭommission on Nomenclature of Organic ChemistryĮXTENSION OF RULES A-1.1 AND A-2.5 CONCERNING NUMERICAL TERMS USED IN ORGANIC CHEMICAL NOMENCLATURE Table: Common Polyatomic ion the topic of acids and polyatomic ions, there is nomenclature of aqueous acids. To correctly specify how many oxygen atoms are in the ion, prefixes and suffixes are again used. This occurs because the number of oxygen atoms are increasing from hypochlorite to perchlorate, yet the overall charge of the polyatomic ion is still -1. 
(Usage of this example can be seen from the set of compounds containing Cl and O) Increasing oxidation state of the nonmetal Hypo chlorite chlorite chlorate per chlorateĪs indicated by the arrow, moving to the right, the following trends occur: \): Slide showing periodic trends for nomenclature of oxyanions of halogens.įigure 2.7.4 Shows several types of polyatomic anions, name 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
